(As of April 2026)
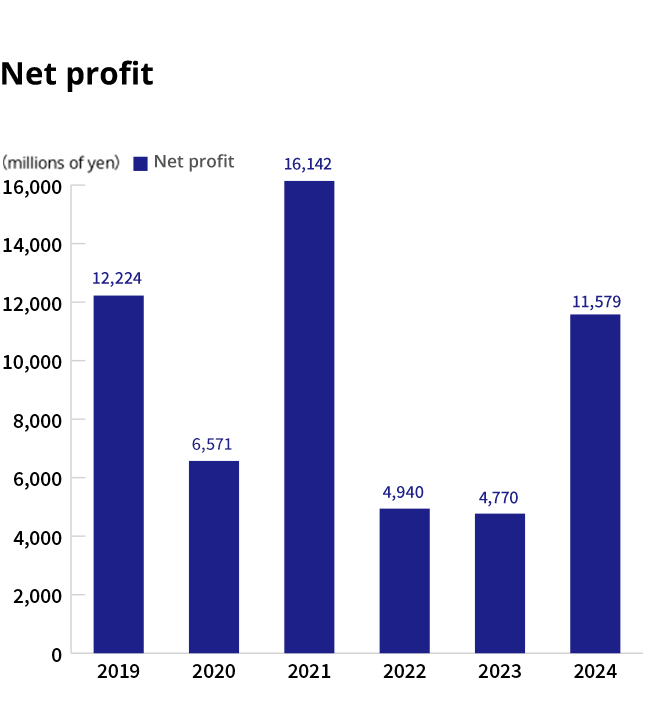
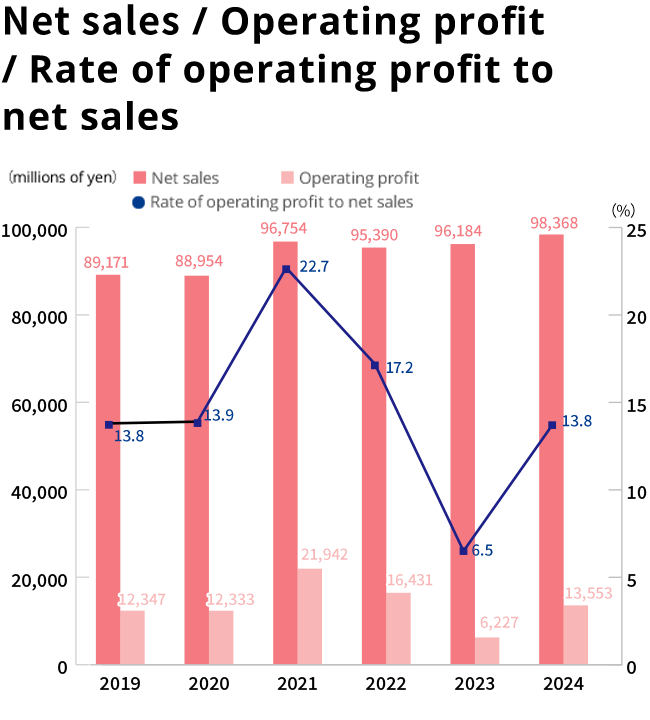
Financial Highlights(consolidated)
-
*New products included in the ratio of new product sales:
The following prescription drugs launched since the fiscal year ended September 2015 BEPIO Gel, BEPIO Lotion, BEPIO Wash Gel, Epiduo Gel, Mitchga Syringes, Mitchga Vials, Renail Gel, Amenalief Tablets, Rozex Gel, Rapifort Wipes, Zebiax Lotion, Zebiax Oily Cream, Comclo Shampoo, Cosentyx for s.c. injection syringe, Cosentyx for s.c. injection pen, Hemangiol Syrup for Pediatric, Marduox Ointment